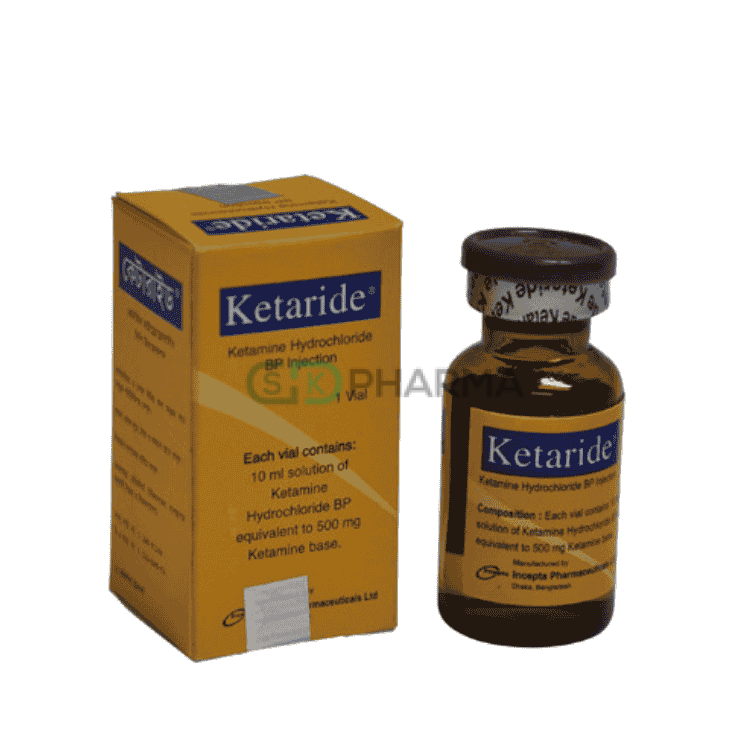
Ketaride Injection 50 mg/ml (Ketamine Hydrochloride)
-
৳5.64
৳6.00 -
৳8.46
৳9.00 -
৳32.90
৳35.00 -
৳9.26
৳10.07
Reviews & Ratings
Ketaride 50 mg/ml is a potent intravenous anesthetic agent designed for the induction and maintenance of general anesthesia. It is particularly useful for short diagnostic or surgical procedures that do not require skeletal muscle relaxation, but it can also be employed in combination with other anesthetic agents for longer or more complex procedures. Ketaride provides rapid, reliable anesthesia while maintaining hemodynamic stability, making it a preferred choice for adults and pediatric patients in controlled clinical settings.
রেজিস্টার্ড চিকিৎসকের পরামর্শ অনুযায়ী ঔষধ সেবন করুন
Therapeutic Class
General (Intravenous) Anesthetic
Pharmacology
Ketaride is a noncompetitive N-methyl-D-aspartate (NMDA) receptor antagonist that blocks the excitatory neurotransmitter glutamate. By acting directly on the cerebral cortex and limbic system, it produces a cataleptic-like state in which the patient becomes dissociated from their surroundings. This unique mechanism provides effective anesthesia without the need for high doses of other agents.
The drug has a rapid onset of action, and its effects can be precisely titrated according to the patient’s anesthetic requirements. Ketaride also exhibits analgesic properties, which contribute to reduced perioperative pain.
Indications
Ketaride 50 mg/ml is recommended for:
-
Sole anesthetic agent: For diagnostic or surgical procedures not requiring skeletal muscle relaxation. Best suited for short-duration procedures.
-
Induction of anesthesia: Prior to administration of other general anesthetic agents.
-
Supplement to low-potency agents: Enhances effects of agents like nitrous oxide for balanced anesthesia.
Dosage and Administration
Adults:
-
IV Induction: 1–4.5 mg/kg
-
IM Induction: 6.5–13 mg/kg
-
IV Infusion: 1–2 mg/kg at 0.5 mg/kg/min
-
Maintenance: Adjust dose according to anesthetic needs; increments of 50–100% of induction dose may be repeated.
-
Rapid Sequence Intubation: 2 mg/kg IV
Pediatrics:
-
IV Induction: 1–2 mg/kg (range: 4–13 mg/kg IM)
-
Maintenance: 0.01–0.03 mg/kg/min continuous IV infusion
Administration Guidelines:
-
Administer slowly over 60 seconds to minimize respiratory depression and cardiovascular effects.
-
Dilute if necessary with sterile water, normal saline, or 5% dextrose.
-
For infusion, reconstitute to 1 mg/ml as instructed.
Precautions and Warnings
-
Contraindicated in severe cardiovascular disease, uncontrolled hypertension, or increased intracranial/intraocular pressure.
-
Caution in patients with cardiac compromise, head injuries, seizures, psychiatric disorders, or hepatic/renal dysfunction.
-
May interact with barbiturates, narcotics, CNS depressants, neuromuscular blockers, thyroid hormones, and antihypertensives.
-
Monitor for emergence reactions such as hallucinations, confusion, or irrational behavior.
Side Effects
-
Cardiovascular: Hypertension, hypotension, tachycardia, bradycardia, arrhythmias
-
Respiratory: Apnea, laryngospasm, respiratory depression
-
Neurological: Increased muscle tone, nystagmus, diplopia, vivid dreams, hallucinations
-
Gastrointestinal: Nausea, vomiting, hypersalivation
-
Miscellaneous: Transient rash, injection site pain, cystitis, raised intracranial and intraocular pressure
Pregnancy and Lactation
-
Pregnancy: Not recommended; safety not established
-
Lactation: May be excreted in breast milk; breastfeeding should be discontinued during use
Storage
Store at 20–25°C, protected from light. Keep out of reach of children.
Related Products
Zinc-R Tablet 20 mg (Zinc Sulfate Monohydrate)
Fenat Tablet 1 mg (Ketotifen Fumarate (Oral))
Ticaflow Tablet 60 mg (Ticagrelor)
Intasone Tablet 10 mg (Hydrocortisone)
Filgrast Injection 300 mcg/0.5 ml (Filgrastim)
Nyclobate Shampoo 0.05% (Clobetasol Propionate)
Product Queries (0)
Login Or Registerto submit your questions to seller
Other Questions
No none asked to seller yet
-
৳5.64
৳6.00 -
৳8.46
৳9.00 -
৳32.90
৳35.00 -
৳9.26
৳10.07