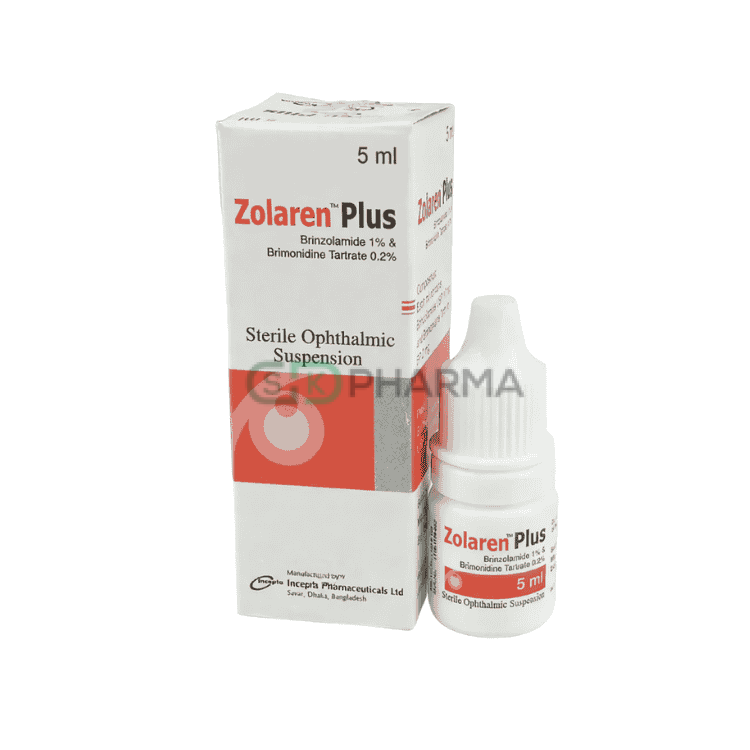
Zolaren Plus Suspension 1%+0.2% (Brinzolamide + Brimonidine Tartrate)
-
৳5.64
৳6.00 -
৳8.46
৳9.00 -
৳32.90
৳35.00 -
৳92.63
৳100.68
Reviews & Ratings
Zolaren Plus Suspension 1%+0.2% combines Brinzolamide and Brimonidine Tartrate and is indicated for the treatment of elevated intraocular pressure (IOP) in patients with ocular hypertension or open-angle glaucoma. Elevated IOP is a major risk factor for optic nerve damage and visual field loss. This dual-action suspension is particularly beneficial in patients whose intraocular pressure is not adequately controlled with monotherapy or who require additional IOP reduction.
Zolaren Plus is useful for:
-
Patients with glaucoma requiring more than one mechanism of IOP reduction
-
Individuals intolerant or insufficiently responsive to other topical glaucoma medications
-
Combination therapy with other ophthalmic agents for enhanced IOP control
By lowering IOP through two complementary mechanisms, Zolaren Plus helps reduce the risk of optic nerve damage and preserves visual function.
Pharmacology
Zolaren Plus contains two active ingredients:
-
Brinzolamide 1% – a carbonic anhydrase inhibitor that decreases aqueous humor production by inhibiting carbonic anhydrase in the ciliary body. Reduced aqueous humor formation leads to lower intraocular pressure.
-
Brimonidine Tartrate 0.2% – an alpha-2 adrenergic agonist that reduces IOP through dual action: it decreases aqueous humor production and increases uveoscleral outflow. Brimonidine also has neuroprotective properties that may help protect the optic nerve from glaucomatous damage.
The combination of these two agents provides additive IOP-lowering effects, achieving better control than either agent alone. Minimal systemic absorption reduces the risk of systemic side effects.
Dosage & Administration
-
Recommended Dose: One drop in the affected eye(s) two times daily.
-
If additional ophthalmic medications are used, administer them at least 5–10 minutes apart to prevent washout and maintain effectiveness.
-
After instillation, patients should gently close the eyes and apply pressure to the inner corner (nasolacrimal duct) for approximately one minute to minimize systemic absorption.
-
Regular monitoring of intraocular pressure is advised to evaluate therapeutic response.
Contraindications
Zolaren Plus Suspension is contraindicated in:
-
Patients with hypersensitivity to Brinzolamide, Brimonidine, other sulfonamides, or any component of the formulation
-
Individuals with severe renal impairment (Brinzolamide) or severe cardiovascular disease (Brimonidine)
-
Children under 2 years of age due to insufficient safety data
-
Patients taking monoamine oxidase inhibitors (MAOIs) due to potential systemic interactions with Brimonidine
Side Effects
Common ocular side effects include:
-
Mild stinging, burning, or discomfort upon instillation
-
Blurred vision or temporary visual disturbances
-
Eye redness or hyperemia
-
Foreign body sensation
Systemic effects may include:
-
Fatigue, dizziness, or dry mouth (mostly due to Brimonidine)
-
Rare allergic reactions, including swelling or itching
Most adverse effects are mild and resolve with continued therapy. If severe reactions occur, treatment should be reassessed.
Precautions & Warnings
-
Use with caution in patients with mild to moderate kidney disease, cardiovascular disorders, or low blood pressure.
-
Avoid contact lenses during treatment if eye irritation occurs.
-
Monitor patients for signs of hypotension, bradycardia, or CNS depression.
-
Do not use concurrently with other alpha-2 adrenergic agonists without medical supervision.
Therapeutic Class
Ophthalmic Carbonic Anhydrase Inhibitor + Alpha-2 Adrenergic Agonist
Storage Conditions
Store Zolaren Plus Suspension 1%+0.2% in a cool, dry place, protected from light. Keep the bottle tightly closed and out of reach of children.
Related Products
Ziton Syrup 10 mg/5 ml (Zinc Sulfate Monohydrate)
Imnoc Tablet 25 mg (Imipramine Hydrochloride)
Palnox Tablet 0.5 mg (Palonosetron)
BetriXa Capsule 40 mg (Betrixaban)
Ovulet Tablet 100 mg (Clomiphene Citrate)
Elivier Tablet 500 mg (Nabumetone)
Vibose Tablet 0.2 mg (Voglibose)
Product Queries (0)
Login Or Registerto submit your questions to seller
Other Questions
No none asked to seller yet
-
৳5.64
৳6.00 -
৳8.46
৳9.00 -
৳32.90
৳35.00 -
৳92.63
৳100.68





![Lifil-E Capsule 400 mg (Vitamin E [Alpha Tocopherol Acetate])](https://skpharma.com.bd/public/uploads/all/iFE69HqHujnjZZbx61PfmcapT1ZsQpwqBHxy0BQg.png)